|
Written by: Huiyu Sheng (MS student, St. Leger Lab) & Zac Lamas (PhD student, vanEngelsdorp Lab) Fungi are more than decomposers. They can infect many different organisms, including insects. It is due to the fact that fungi have the ability to infect insects that humans have a long history of using them for the biological control of pests. For instance, Beauveria bassiana was first identified as a silkworm pathogen in 1815. In 1879, Elie Metchnikoff, a Russian zoologist suggested using Metarhizium anisopliae to control beetles of agricultural crops.  Fig 1. A zombie carpenter ant which was killed by a parasitic fungus. Image via David P. Hughes, Penn State University. Fig 1. A zombie carpenter ant which was killed by a parasitic fungus. Image via David P. Hughes, Penn State University. In fact, many fungi have evolved to be efficient insect killers. Fungal spores will germinate when they land on an insect’s cuticle, and slowly burrow their way through the insect’s protective cuticle. Once the fungus reaches the insect’s “blood”, it will reproduce using the nutrients in it. In this way fungi have become excellent insect pathogens. Some fungi have evolved to be generalist pathogens, killing many different species of insects. Other fungi are specialists that have a narrow range of insects they can target, for example, some fungi kill a single species of ant (Fig 1). Brian Lovett, a recent PhD graduate at the University of Maryland, has studied how fungi can be used to reduce malaria cases in sub-Saharan Africa. Is it really possible the insect killing abilities of fungi can be used to specifically target mosquitos that vector malaria? Brian Lovett has been developing a transgenic strain of a fungus capable of reducing mosquito populations. Mosquitos, and the diseases they vector, are still a worldwide issue. Over 200 million people contract malaria each year. These cases largely occur in sub-Saharan Africa, and in recent years incidence of malaria has plateaued, instead of diminished. This is largely because pyrethroids, the class of insecticides largely used for mosquito control, are losing their efficacy. Without new methods to control mosquito populations in malaria endemic areas, the risk of malaria incidence rising is a real threat. Recent advances have allowed scientists to modify fungi that can be used for insect control. The St. Leger lab at the University of Maryland developed a transgenic bioinsecticide by using Hybrid toxin, originally derived from the venom of the Australian Blue Mountains funnel-web spider, and inserted it into the genome of Metarhizium pingshaense. Because this toxin has several targets in insect neurons, there is a low chance of mosquitoes developing resistance to it [2]. The team also found Hybrid-expressing fungus was more effective at killing mosquitos. 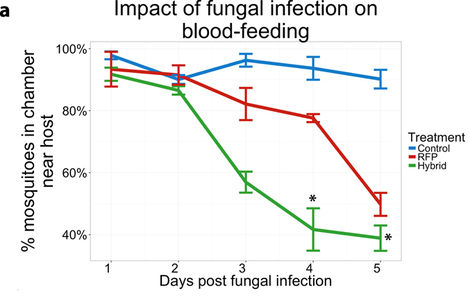 Fig 2. Impact of fungal infection on blood-feeding. Hybrid fungus can significantly reduce the blood feeding interest of mosquito as soon as the neurotoxin was expressed(day3). Fig 2. Impact of fungal infection on blood-feeding. Hybrid fungus can significantly reduce the blood feeding interest of mosquito as soon as the neurotoxin was expressed(day3). With this foundation and funding from the National Institutes of Health, they collaborated with scientsits in Burkina Faso to conduct semi-field experiments with the transgenic fungus in a purpose-built MosquitoSphere in Soumousso, a rural village where malaria is endemic. This international team tested the mosquito killing potential of the novel fungus and compared it to the unmodified fungus. They placed a black cotton cloth which had been impregnated with fungal spores in an experimental hut with a calf as a blood source for mosquitos. Later they collected all the mosquitos in each treatment group (Hybrid fungus, wild-type fungus and a control group with no fungus). Mosquitos that landed on the cloth with Hybrid fungus were dead within 14 days, which was faster than the mosquitos treated with unmodified fungal spores. Then, they tested if the impact of Hybrid fungus could affect the development of the next generation of mosquitos. They applied the fungi in the same way and found the Hybrid-expressing fungus can collapse a population in two generations, while the populations of mosquitos treated with the control and wild-type fungus rose. In order to know if the mosquitos still were interested in human blood once they have been infected by the fungus, they conducted an experiment by using guinea pig as the an attractant [3]. He found that the Hybrid fungus can significantly reduce the blood-feeding interest of mosquitos as soon as the neurotoxin was expressed (Fig 2). Finally, they tested if the Hybrid fungus can affect mosquitoes over several generations. They continuously checked the mortality of these mosquitos during the rainy season. Even though the cloth with Hybrid fungus gradually lost its virulence over time in field conditions, it still effectively controlled the mosquitos for 6 weeks. This team was conscientious of socio-economic needs in their experimental design. For the project to be successful, applications would need to be cost effective. The cotton cloths used in their trials and the sesame oil are both readily available and inexpensive. Brian believes using the transgenic Hybrid strain with a single re-application would remain effective throughout the whole rainy season. There are many remaining steps before field application of such a transgenic fungus can occur. The next step for this group is to earn stakeholder approval for field testing. This team needs to work directly with regulatory bodies in Burkina Faso and with community members before they can attempt a field trial. Field trails assessing ecological risk and demonstrating successful entomological, then epidemiological endpoints are essential before the sale and distribution of transgenic fungus for biocontrol. References: [1] Liao X, Fang W, Lin L, Lu H-L, Leger RJS (2013) Metarhizium robertsii Produces an Extracellular Invertase (MrINV) That Plays a Pivotal Role in Rhizospheric Interactions and Root Colonization. PLoS ONE 8(10): e78118. https://doi.org/10.1371/journal.pone.0078118 [2] Lovett, B., Bilgo, E., Millogo, S. A., Ouattarra, A. K., Sare, I., Gnambani, E. J., … St. Leger, R. (2019, May 31). Transgenic Metarhizium rapidly kills mosquitoes in a malaria-endemic region of Burkina Faso. Science, 894–897. [3] Bilgo, E., Lovett, B., Fang, W., Bende, N., King, G. F., Diabate, A., & St Leger, R. J. (2017). Improved efficacy of an arthropod toxin expressing fungus against insecticide-resistant malaria-vector mosquitoes. Scientific reports, 7(1), 3433. doi:10.1038/s41598-017-03399-0 Authors: Huiyu Sheng is a second year master’s student in the St. Leger lab, studying on entomo-pathogenic fungi and their relationship with both plants and insects. Zac Lamas is a second year PhD student studying the feeding behavior of Varroa destructor mites. Comments are closed.
|
Categories
All
Archives
June 2024
|
Department of Entomology
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290

 RSS Feed
RSS Feed




