Hitting the Weak Spot: Biologically based pest management of the Brown Marmorated Stink Bug10/8/2018
 Figure 1. BMSB adult Figure 1. BMSB adult Written by: Maggie Lewis. Maggie is a second year PhD student in the Hamby lab who is currently studying the interactions between spotted wing drosophila and yeast and fungal microbes. In agriculture, understanding an insect’s biology is a crucial aspect for developing and improving sustainable agricultural pest management programs. Knowledge of basic aspects of an insect’s life history, including its phenology, its behavioral ecology, and its interactions with the environment, provide clues that can help growers identify and exploit that pest’s weakness. Dr. Anne Nielsen, an associate professor in the Department of Entomology at Rutgers University, is currently studying how we can use this biologically based approach to improve management of Halyomorpha halys, more commonly known as the brown marmorated stink bug (BMSB). 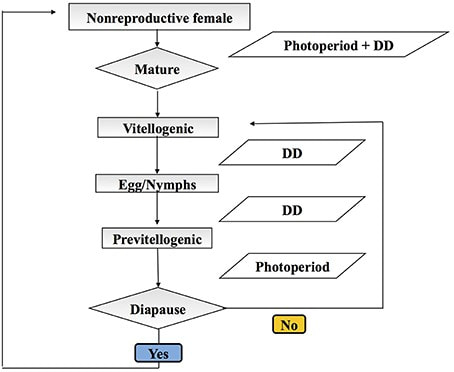 Figure 2. Diagram of the BMSB model development. Degree days accumulation and photoperiod were measured at various developmental timepoints (indicated by grey boxes). Image from Nielsen et al. 2016. Figure 2. Diagram of the BMSB model development. Degree days accumulation and photoperiod were measured at various developmental timepoints (indicated by grey boxes). Image from Nielsen et al. 2016. BMSB is an invasive stink bug that is native to China and is perhaps best known for its tendency to overwinter in human structures. However, in addition to being a major nuisance for homeowners, BMSB is also a key agricultural pest in many cropping systems. In the eastern United States, BMSB was first detected in the 1990s, and was elevated to major pest status in the 2000s. Though populations of BMSB have recently begun declining in the eastern US (for reasons not entirely understood), this pest has continued to spread and is a still a major problem in many western states. Part of the difficulty in managing BMSB stems from the fact that it has an extremely broad host range that includes agricultural crops like apples, peaches, and soybeans. BMSB is also highly mobile, and both the adult and immature stages are known to switch between hosts rapidly. Therefore, management programs for BMSB typically require a landscape level approach. One pillar of Dr. Nielsen’s work is BMSB phenology, or modeling of the timing of key events in the pest’s life history. Most insect phenology models are developed using a biofix (a defined biological indicator or event that serves as the starting point for the model) followed by measurements of heating and cooling. However, these degree-day based models often ignore critical life-history functions (like diapause) that may not be informed by temperature. To improve the accuracy of these temperature-based models, Dr. Nielsen decided to take an agent based modeling approach, which incorporates temperature as well as various physiological aspects of BMSB’s biology, such as when they overwinter or when females break diapause to become reproductively mature. To develop these models, Dr. Nielsen collected non-reproductive female BMSB from 8 distinctive geographic locations. She tracked the development of these females; they eventually matured into reproductive females and produced eggs that then matured into a second generation of adults (Figure 2). The newly emerged females in this second generation could then either enter diapause (and suspend reproduction) or become reproductive adults. Using this data, Dr. Nielsen was able to record the period of time (as degree days) between these key physiological events, the photoperiod (length of day) at which diapause occurred, and survivorship at each key phenological stage (Figure 2). These studies were then incorporated with other work into a larger, cumulative model of BMSB’s phenology, developed in collaboration with Dr. Shelby Fleischer and Dr. Shi Chen (Nielsen et al. 2016).  Figure 3. Trissulcus japonicas, an egg parasitoid of BMSB Figure 3. Trissulcus japonicas, an egg parasitoid of BMSB Surprisingly, the resulting phonological model predicted that BMSB is bivoltine (or goes through two generations annually), contradicting the widespread belief that BMSB was univoltine. Through subsequent model validation, Dr. Nielsen determined that BMSB’s F1 generation occurs in early spring in wooded areas. In the mid-Atlantic region or other northern states, this F1 generation is fairly small; that, combined with the fact that it occurs before stink bugs move into agricultural fields, had prevented researchers from detecting the presence of a separate F1 generation. Instead, the large annual peak of BMSB observed in agricultural fields is actually a second, or F2, generation. While this bivoltine phenological pattern appears to be consistent throughout the United States, the proportion of BMSB that come from each generation varies considerably across geographic regions, depending on both the temperature and the length of day within a given region. Using this model, researchers can now begin to predict dispersal and timing of key events within BMSB’s life cycle. Dr. Nielsen is currently collaborating with researchers at other universities to develop an online database that can be updated in real time and may eventually be incorporated into trapping networks. The information that this model generates, including when BMSB populations will peak, the number of generations that occur annually, and the periods of peak oviposition, can help inform key pest management decisions, such as when to time pesticide sprays or release biocontrol agents. Another aspect of Dr. Nielsen’s research takes a systems–level approach to agricultural pest management in tree fruit. Since BMSB was first introduced to the United States, there has been almost a four-fold increase in pesticide sprays in tree fruit, representing almost a complete reversal of previously established pest management programs. To bring this pesticide usage down, Dr. Nielsen developed an IPM-CPR program (Integrated Pest Management – Crop Perimeter Restructuring) in peaches and apples. BMSB is frequently described as an edge pest, which means that it primarily colonizes the border of an agricultural field. Exploiting that behavior, a key aspect of Dr. Nielsen’s IPM-CPR program is to only spray pesticides around the orchard border. In the unsprayed center region, Dr. Nielsen then brought back previously used IPM tactics, including mating disruption to target other key fruit pests (such as the oriental fruit moth) and ground cover management. The latter tactic, which involves removed removing flowering weeds attractive to pollinators and lygus bugs, helps ensure that bees and other beneficial insects were not present in the orchards when pesticides are sprayed. Working on commercial fruit farms, Dr. Nielsen evaluated this system’s efficacy over three years (Blaauw et al. 2014). In general, she found that growers who utilized IPM-CPR significantly reduced their insecticide application per acre. In the first year of the study, there were significantly lower rates of insect injury to fruit in the IPM blocks compared to the conventional (full-spray) treatments. In the following two years, she observed no significant differences between the two treatments, indicating that both approaches were equally effective at managing BMSB and other fruit pests. More recent studies also suggest that IPM-CPR may benefit the natural enemy community. At some field sites, Dr. Nielsen has observed increased biodiversity of generalist BMSB predators, such as coccinellids and nabid bugs. However, the results have not been consistent across all IPM blocks, indicating that future studies will be needed to tease apart these treatment effects. But, in an ongoing study, Dr. Nielsen has found that an exotic egg parasitoid of BMSB, Trissulcus japonicas (Figure 3), will disperse into IPM blocks to parasitize BMSB. Taken together, Dr. Nielsen’s work highlights the importance of taking the time to know a pest’s biology. Understanding its life history and identifying the weak points can help researchers develop novel management tactics. As we see in the IPM-CPR program, these tactics can be successfully employed to improve sustainable pest management programs. References Blaauw, B. R., Polk, D., and Nielsen, A. L. 2014. IPM-CPR for peaches: incorporating behaviorally-based methods to manage Halyomorpha halys and key pests in peach. Pest Manag Sci. Nielsen, A. L., Chen, S., and Fleischer, S. J. 2016. Coupling Developmental Physiology, Photoperiod, and Temperature to Model Phenology and Dynamics of an Invasive Heteropteran, Halyomorpha halys. Front. Physiol. Comments are closed.
|
Categories
All
Archives
June 2024
|
Department of Entomology
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290

 RSS Feed
RSS Feed




