PICK LAB OBSERVATIONS OFFER CLUES INTO UNDERSTANDING HOW DIFFERENT MODES OF SEGMENTATION EVOLVE12/15/2021
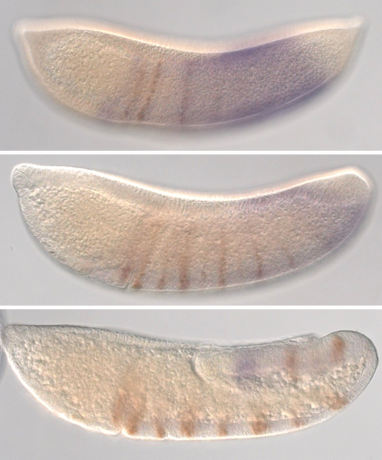 Image caption- In the mosquito Anopheles stephensi, expression of segment marker gene Engrailed (brown) is activated in a wake of a wave of opa (purple) expression, which times segment specification during Drosophila development. Image caption- In the mosquito Anopheles stephensi, expression of segment marker gene Engrailed (brown) is activated in a wake of a wave of opa (purple) expression, which times segment specification during Drosophila development. Arthropods are famous for their segmented body plans, which contribute to their vast diversity through modular addition of appendages, wings and more. Segments are patterned early in embryonic development, and this process has been intensively studied in the fruit fly, Drosophila melanogaster. Drosophila is a premier model system, owing to ease and speed of culture and abundant genomic and molecular tools. However, it’s segmentation process is seemingly unlike that of most arthropods. Drosophila establishes all its segments at the same time, whereas other species specify segments one at a time over a long growth period. It has long been assumed that distantly related flies, such as mosquitoes, make this segments the same way that Drosophila do. However, while exploring the loss of an important segmentation gene from mosquito genomes, the Pick lab noticed that mosquito segmentation appeared to represent an intermediate, blending aspects of simultaneous and sequential segmentation. In their new work, published in JEZ-B, Alys Cheatle Jarvela, Catherine Trelstad, and Leslie Pick characterize this intermediate segmentation mode in the mosquito, Anopheles stephensi, and show that it is likely controlled by a progressive wave of developmental regulatory gene expression. These observations offer clues into understanding how different modes of segmentation evolve and have opened new lines of research into segment patterning in the Pick lab. Comments are closed.
|
Categories
All
Archives
June 2024
|
Department of Entomology
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290

 RSS Feed
RSS Feed




