[Seminar Blog] The Battle for Blood - How Mosquito Salivary Proteins Facilitate Blood Feeding10/4/2023
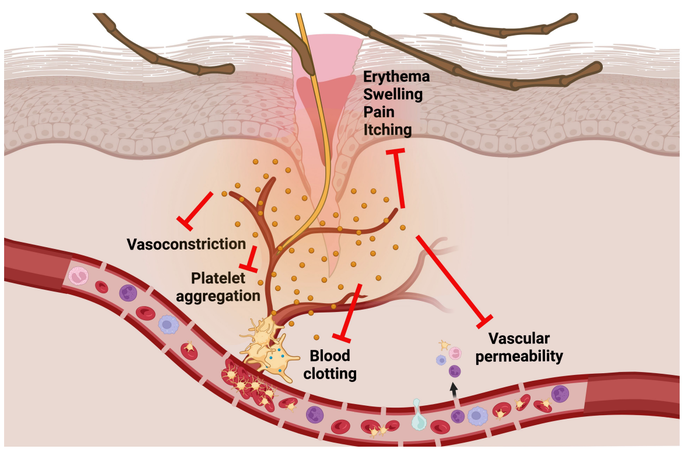
written by: Jenan El-Hifnawi, Ben Gregory, and Minh Le Imagine it’s a hot summer day and you’re slurping down a strawberry milkshake. Suddenly you’re interrupted by a pesky little ice chip clogging your straw. While this experience is all too familiar among your fellow humans, you may not have guessed that mosquitoes share a similar struggle! Mosquitoes use their long sucking mouthparts (called a proboscis) to feed on our blood in a similar way to a human using a straw. To a mother mosquito, drinking your blood is a vital step in building her eggs, which she will later lay in the water. However, while she slurps up your blood, your body fights back against the wound and actively tries to stop the bleeding. Your blood vessels constrict to stop the flow of blood, a clot forms at the site of the puncture, and the area around the bite becomes inflamed. This response is called hemostasis. While we humans have the luxury of being able to blow that ice chip out of our straw, mosquitoes have had to evolve a more subtle way to finish their milkshake. To keep your blood flowing, mosquitoes secrete a cocktail of specialized proteins into your bloodstream through their saliva. These tiny biomechanical machines prevent clotting and constriction of blood vessels, making it much harder for our bodies to minimize blood loss. These proteins also decrease the permeability of our blood vessels, interfering with the body's ability to deliver its itch-causing allergy response. (The itchiness and irritation begin only after the mosquito has taken her blood meal and flown away.) There are somewhere between 150 and 200 different proteins in this salivary cocktail, most of which have completely unknown functions. If we can understand the complex biochemistry of these proteins, we can take one step closer to preventing and mitigating mosquito-borne illnesses. That’s where the National Institute of Health’s Chief of Molecular Entomology, Dr. Eric Calvo comes in. His lab identified a group of proteins in the mosquito salivary cocktail known as the D7-related proteins. One protein in this group, D7L1, is of particular interest to Dr. Calvo’s team. In a series of experiments, they found that the D7L1 protein binds to and disables an important molecule for blood clotting called ADP. This suggested that D7L1 prevents clotting, but they wanted more evidence.
In an elegant experiment, Dr. Calvo’s lab used CRISPR/Cas9 gene editing technology to genetically modify Aedes aegypti (Yellow fever mosquito) and break two of their D7-related proteins: D7L1 and D7L2. When given the opportunity to feed on mice, mosquitoes with broken copies of these proteins repeatedly stabbed at different locations with their proboscis for minutes at a time, and often never settled down on a blood vessel to start taking blood. On the other hand, unmodified mosquitoes with working D7-related proteins managed to find a blood vessel and start feeding in a matter of seconds. This longer “probing time” in the genetically modified mosquitoes suggests that without D7-related proteins, mosquitoes are unable to prevent clotting, and therefore fail to ingest blood. In a follow-up experiment, Dr. Calvo’s lab tested the same genetically modified mosquitoes on mutant mice that had their clotting and vasoconstriction abilities disabled. The result was the astonishing return of the mosquitoes’ fast probing time. It appears that since the hemostasis functions in mice were removed, the D7L1 and D7L2 proteins in mosquitoes were no longer needed and the mosquitoes could slurp without worrying about clots. So, could we disable these genes in wild mosquitoes and give ourselves more time to slap them as they frantically stab at our skin, looking for a vessel that won’t clot? While it’s easy to imagine how this would severely hurt a mosquito’s chance at survival, the impacts of probing time on pathogen transmission are more nuanced. Pathogen transmission tends to occur during the injection of the proboscis, not while the mosquito is feeding on your blood. Therefore, the more time a mosquito spends probing, the higher the chance of transmitting these deadly agents, so we might have to look elsewhere for solutions. While beating the bites may be on the forefront of Maryland residents' minds this summer, Dr. Calvo’s work creates opportunities for cutting edge discoveries on many fronts. First and foremost, this research has pressing implications for our understanding of how mosquitoes’ vector some of the world’s deadliest diseases, like malaria and West Nile virus. Given D7 proteins’ important role in modulating transmission of these pathogens, it could be a viable target for vaccine development. Furthermore, Dr. Calvo and his team have identified compounds which can prevent blood clotting through unique mechanisms - a discovery which could be harnessed to manufacture drugs with the same goal. Learn more about Dr. Eric Calvo’s work through his recent publications: Smith LB, Duge E, Valenzuela-León PC, Brooks S, Martin-Martin I, Ackerman H, Calvo E. Novel salivary anti-hemostatic activities of long-form D7 proteins from the malaria vector Anopheles gambiae facilitate hematophagy. J Biol Chem. 2022 Apr 20:101971. Martin-Martin I, Valenzuela Leon PC, Amo L, Shrivastava G, Iniguez E, Aryan A, Brooks S, Kojin BB, Williams AE, Bolland S, Ackerman H, Adelman ZN, Calvo E. Aedes aegypti sialokinin facilitates mosquito blood feeding and modulates host immunity and vascular biology. Cell Rep. 2022 Apr 12;39(2):110648. Student Author Biographies: Jenan El-Hifnawi: Jenan is a first-year Masters student in the Espíndola Lab. Her research seeks to explore the impacts of Ice Age glaciations on the diversification and range of several South Andean bee species using phylogeographic methods. Ben Gregory: Ben is a second-year PhD student in the Fritz lab. He is interested in using spatial and genomic modeling techniques to map gene flow, hybridization, and local adaptation within the Culex pipiens assemblage along urbanization gradients in the United States. Minh Le: Minh is a second-year PhD student in the Pick lab. He is interested in the evolution of transcription factors involved in insect segmentation. Comments are closed.
|
Categories
All
Archives
June 2024
|
Department of Entomology
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290


 RSS Feed
RSS Feed




