[Seminar Blog] USDA Protects US Agriculture Using Sterilized and Genetically Engineered (GE) Insects2/3/2022
Written by: Krisztina Christmon and Veronica Yurchak From its first use in the 1950’s through present day, scientists have been trying to manage insect pests using the sterile insect technique (SIT). Today, USDA ‘s Animal and Plant Health Inspection Service (APHIS) - Plant Protection and Quarantine unit uses SIT as part of their Fruit Fly Exclusion and Detection Program, the goal of which is to protect US Agriculture from potentially invasive and damaging exotic fruit fly species. Dr. Corey Bazelet is responsible for coordinating this fruit fly program and presented her work as part of the University of Maryland’s Safeguarding American Agriculture seminar series. Differing from common domestic fruit flies commonly found annoying homeowners in summer kitchens, many exotic fruit flies are among the most destructive agricultural pests worldwide. As such, they pose a significant risk to American agriculture. In addition to reducing yield and fruit quality, fruit flies also interfere with international trade. Increased monitoring and pre-import inspections, additional offshore pest control treatments like fumigation or irradiation, as well as potential port entry denials can all disrupt trade for commodities subject to fruit fly infestations. As such, the major activities of APHIS’ domestic fruit fly program focus on the exclusion of exotic fruit fly species; specifically, the Medfly and Mexfly from the Mediterranean area and Mexico, respectively (Figure 1). The primary tactic used in the fight against fruit fly establishment is the release of fruit flies sterilized using SIT. 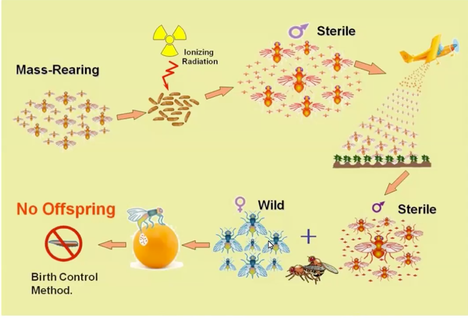 Figure 2. Depicts the process of creating and releasing sterilized insects as part of the SIT. Image Source: Dr. Corey Bazelet. Figure 2. Depicts the process of creating and releasing sterilized insects as part of the SIT. Image Source: Dr. Corey Bazelet. Essentially a form of “fly birth control”, SIT requires facilities capable of rearing millions of fruit flies in a factory-like setting. These fruit flies are then separated by sex, and male fruit flies are exposed to radiation, typically during the pupal stage, which results in sterile adult fruit flies. (For a video description of the sterile insect technique click here!) After rearing, sterile adults are released at targeted locations where they can encounter and mate with exotic fruit flies. Importantly, because the flies are sterilized before their release, the interaction does not result in fruit fly offspring. The overwhelming number of sterile adult males released ensures that no invading females will encounter an invading, non-sterilized male (Figure 2). APHIS operates two preventative release fruit fly programs within three states: the Mediterranean Fruit Fly Program, which releases of sterile Mediterranean fruit flies in California (300 million flies per week) and Florida (50 million per week), and the Mexican Fruit Fly Program, which releases of sterile Mexican fruit flies in Texas (150 million per week) and Northern Mexico (20 million per week). By releasing such large numbers, APHIS aims to ensure that any potential exotic fruit flies that have evaded port commodity inspections will be overwhelmed by the sterile male adults, and thus, are unable to establish a population. Future directions for this program aim to improve the sterile insect technique by separating flies in rearing facilities as eggs, rather than pupae, and, eventually, using gene editing technologies to produce only male eggs, eliminating the need to separate out female flies at a later stage. Although the sterile insect technique has been successful at preventing various pest infestations within the US, the continuous rearing and release process is quite labor intensive and more easily done for some pest species than others. For many insect pests, the prospect of control through genetic engineering offers more promising results. Dr. Lisa Knolhoff of the USDA APHIS Biotechnology Regulatory Services presented during the second portion of the seminar on the use of genetically engineered (GE) insects, with a focus on the diamondback moth as a model system to study how agricultural pests adapt to management practices. Dr. Knolhoff’s work provides an example of using genetic engineering technologies to accomplish similar goals as SIT. The diamondback moth (Figure 3)is a major pest worldwide, and it has evolved resistance to most major classes of insecticides. It was the first crop pest to evolve resistance to DDT and the first pest to develop all resistance to Bt sprays. DDT was the first modern synthetic insecticides in the 1940s. It was initially very effective, but due to detrimental effects on human health and the environment it was banned in 1972. Bacillus thuringiensis (Bt) has been widely used since 1961 as a pesticide as it is naturally found in soil, but it has never been as effective as some chemical pest control methods. In contrast to chemical pest control which often kill a broad range of insects, genetically engineered pests are species-specific pest control methods. One example of the modified pest biology pest control is the female specific Release of Dominant Lethal (fsRIDL) technology. The RIDL system is a variant of SIT which replaces irradiation by genetically engineered inducible sterilization, an approach offering more flexibility with regards to the time of death, the sex and even the tissues targeted by the sterilizing mechanism. Once this technology was developed in the laboratory for the diamondback moth, the research team did field trials to assess environmental impacts under the guidelines of the National Environmental Policy Act (NEPA). For all field trials that the agencies authorize, regardless of the organism, there are requirements for both in season and post season monitoring to make sure that it does not spread or persist beyond the duration of the trial. At the conclusion of the trial, the area is sprayed with insecticide and tilled to break up the vegetative material and subsequently expose it to winter conditions. Finally, monitoring continues to make sure that no GE diamondback moth individuals remain. It’s important that these technologies are effective at controlling pests, but also to ensure they are not altering other ecological dynamics.
Emerging technologies such as GE both poses challenges and opportunities, and an important aspect of both Dr. Bazelet and Dr. Knolhoff’s jobs is to keep up with them in order to keep US agriculture and the environment safe. References and further reading: Visit the following sources for further information on the pests described in this article as well as information on what you can do to prevent the spread of invasive agricultural pests:
Authors: Krisztina Christmon, PhD student at vanEngelsdorp bee lab studying the host-parasite-pathogen interaction of the honey bee parasite: Varroa destructor. Veronica Yurchak, PhD student in Dr. Hook’s applied ecology lab studying alternative methods for pest control in sweet corn production. Comments are closed.
|
Categories
All
Archives
June 2024
|
Department of Entomology
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290
University of Maryland
4112 Plant Sciences Building
College Park, MD 20742-4454
USA
Telephone: 301.405.3911
Fax: 301.314.9290




 RSS Feed
RSS Feed




